Influenza Ag A+B Test
Quick Details
| Type | Detection Card |
| Used for | Salmonella Typhi Test |
| Specimen | Feces |
| Assy Time | 5-10 minutes |
| Sample | Free Sample |
| OEM Service | Accept |
| Delivery Time | Within 7 working days |
| Packing Unit | 25 Tests/40 Tests |
| sensitivity | >99% |
● Easy to operate, fast and convenient, can read the result in 10 minutes, diversified application scenarios
● Pre-packed buffer, the use of steps more simplified
● High sensitivity and specificity
● Stored at room temperature, valid for up to 24 months
● Strong anti-interference capability
The S.typhi Antigen Rapid Test Cassette (Feces) is a rapid chromatographic immunoassay for the qualitative detection of Salmonella typhi antigens in human feces specimens to aid in the diagnosis of Salmonella typhi infection.Typhoid fever is a life threatening illness caused by the bacterium Salmonella typhi, and was observed by Eberth (1880) in the mesenteric nodes and spleen of fatal cases of typhoid fever.
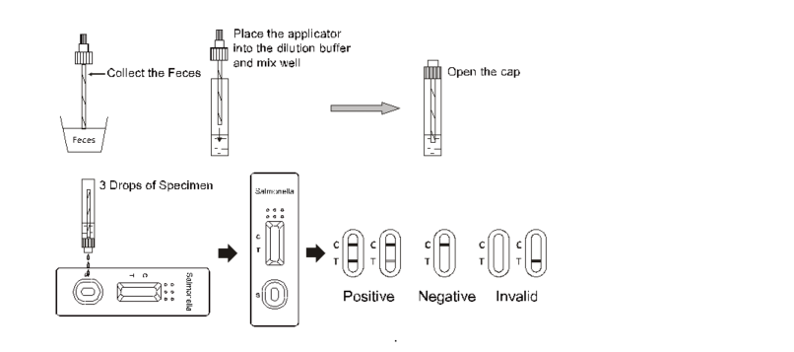
Test Procedure
Allow the test, specimen and/or controls to reach room temperature 15-30℃ (59-86℉) prior to testing.
1.Bring the pouch to room temperature before opening it. Remove the test device from the sealed pouch and use it as soon as possible.
2.Place the test device on a clean and level surface.
3.Holding the sample collection tube upright, carefully take off the tip of collection tube, transfer 3 drops (approximately 100μl) to the specimen well of the test device, then start the timer. See illustration below.
4.Wait for the colored line to appear. Read results at 15 minutes. Do not interpret the result after 20 minutes.
Notes:
Applying sufficient amount of specimen is essential for a valid test result. If migration (the wetting of membrane) is not observed in the test window after one minute, add one more drop of specimen.